Characterization and thermal analysis of metalworking sludge as a partial substitute for clays in ceramic production
Andreia Cristina Brenner Fontana[1]
Alvaro Luiz Mathias[2]
Abstract
This study evaluates the feasibility of using sludge from the metalworking industry (SMWI) as a partial substitute for clays in producing red ceramics for civil construction. The analysis of the white goods production process predicted the components of SMWI, potentially reducing the need for additional chemical analyses. According to the chemical analyses provided, SMWI is a non-hazardous solid waste, but not inert, due to the presence of aluminum, chlorine, and phenols, by Brazilian standards. The oxide composition of SMWI showed compatibility with the studied clays and data from the literature, suggesting its potential as an additive in ceramics. Rich in CaO and Al2O3, SMWI can enhance the mechanical strength of ceramics, although excessive addition may increase water absorption. Loss on ignition tests, thermogravimetry, and SEM-EDS analyses on SMWI and pure and 5% SMWI-added clays revealed the loss of volatile compounds and the formation of thermostable clay minerals. However, its distinct composition, with lower levels of Fe2O3 and SiO2, and the need for adequate homogenization, requires caution to avoid impacts on the final properties of the ceramic products, such as water absorption and mechanical strength. Thus, the use of SMWI as a ceramic additive presents itself as a sustainable and viable alternative, although further studies are necessary to validate its application in compliance with Brazilian technical standards.
Keywords: Sustainable Management; Industrial Sludge; Pollutant Prediction; Waste Valorization; Clay Substitution.
Caracterização e análise térmica de lodo metalmecânico como substituto parcial de argilas na produção cerâmica
Resumo
Este estudo avalia a viabilidade de utilizar o lodo da indústria metalmecânica (SMWI) como substituto parcial de argilas na produção de cerâmicas vermelhas para a construção civil. A análise do processo de produção de eletrodomésticos previu os componentes do SMWI, potencialmente reduzindo a necessidade de análises químicas adicionais. Segundo as análises químicas previstas, o SMWI é um resíduo sólido não perigoso, mas não inerte, devido à presença de alumínio, cloro e fenóis, conforme as normas brasileiras. A composição de óxidos do SMWI mostrou compatibilidade com as argilas estudadas e dados da literatura, sugerindo seu potencial como aditivo em cerâmicas. Rico em CaO e Al2O3, o SMWI pode aumentar a resistência mecânica das cerâmicas, embora a adição excessiva eleve a absorção de água. Ensaios de perda ao fogo, termogravimetria e MEV-EDS no SMWI e em argilas puras e aditivadas com 5% revelaram a perda de compostos voláteis e a formação de argilominerais termoestáveis. No entanto, sua composição diferenciada, com menores teores de Fe2O3 e SiO2, e a necessidade de uma homogeneização adequada exigem cautela para evitar impactos nas propriedades finais das cerâmicas, como absorção de água e resistência mecânica. Portanto, o uso do SMWI como aditivo cerâmico surge como uma alternativa sustentável e viável, mas estudos complementares são necessários para validar sua aplicação conforme as normas técnicas brasileiras.
Palavras-chave: Gestão Sustentável; Lodo Industrial; Previsão de poluente; Valorização de Resíduo; Substituição de argila.
Recebido em: 05/09/2024
Aceito em: 27/09/2024
Publicado em: 01/02/2025
1 Introduction
The following section presents the relevance of using industrial waste, particularly metallurgical sludge, as an alternative raw material in ceramic production. Based on environmental regulations and the growing need for sustainable practices, this study investigates the potential of incorporating such waste into brick manufacturing raw material as a final destination. Through detailed chemical and thermal analyses, the research aims to assess the feasibility of replacing natural clays with metallurgical sludge, highlighting its environmental and economic benefits while addressing key industrial challenges.
The generation of large volumes of urban and industrial waste is a pervasive challenge, often unavoidable but undesirable. Like the European Union Directive 2008/98/EC (EU, 2008), Brazil's National Solid Waste Policy, Law No. 12,305 of August 2, 2010 (BRASIL, 2010), sets up clear guidelines to inform decision-making processes. This legal framework prioritizes actions in the following order: prevention of waste generation, reduction, reuse, recycling, treatment, or, eventually, environmentally final disposal. Within this context, the incorporation of urban solid waste, such as beached sargassum, into the production of construction materials offers a practical pathway to mitigate various environmental impacts associated with land use (Parente et al., 2024).
In a similar context, the treatment of water for human consumption, sewage, and industrial effluent generates sludge through processes such as coagulation, flocculation, and precipitation (Richter, 2001; Metcalf & Eddy, 2015). The chemical and physical properties of sludge vary depending on the characteristics of the water, industrial inputs, and specific treatment processes (Ahmad; Ahmad; Alam, 2016). For instance, the metalworking industry (SMWI) produces predominantly inorganic sludge, primarily composed of aluminum oxide, a byproduct of the coagulants used (Vieira; Silva, 2012). According to Brazil’s National Solid Waste Policy (BRASIL, 2010), this type of solid waste can potentially be treated to recover the coagulant agent for reuse as a contaminant and heavy metal adsorbent in effluent treatment, as well as in various applications such as construction materials, soil additives in agriculture, and as a secondary raw material for cement and brick manufacturing (Ahmad; Ahmad; Alam, 2016). Coagulation sludges that share comparable properties with clays used in ceramic production can partially replace this primary raw material, conserving natural resources (Tartari et al., 2011; Sutcu et al., 2022). This approach significantly mitigates environmental impacts, including soil erosion, water contamination, disruptions to local flora and fauna, deterioration of air quality, noise pollution, and the loss of landscape value due to clay extraction. It also addresses socio-environmental conflicts (López-Juvinao; Torres-Ustate; Moya-Camacho, 2020) and reduces the need for rehabilitating degraded areas (Turrión et al., 2021). Thus, the valorization of coagulation sludge as a partial substitute for clay in ceramic brick production is a sustainable resource management strategy (Ahmad; Ahmad; Alam, 2016) that aligns with recycling principles (EU, 2008; BRASIL, 2010).
Several studies have demonstrated promising results when incorporating various types of sludge into ceramic bricks, including metalworking sludge (Vieira; Silva, 2012), ceramic sludge (Coletti et al., 2016), tannery sludge (Juel; Mizan; Ahmed, 2017; Hasan; Hashem; Payel, 2022), marble sludge (Munir et al., 2018), papermaking and sugar carbonation sludge (Yaras, 2020), dairy factory sludge (Simón et al., 2021), water treatment plant sludge (Oliveira; Holanda, 2008; Tartari et al., 2011; Benlalla et al., 2015; Sutcu et al., 2022), and urban wastewater effluent sludge (Martínez-García et al., 2012; Zhang et al., 2016; Ukwatta et al., 2015; Ukwatta et al., 2016; Ukwatta; Mohajerani, 2017). Red clays, typically used in brick production, contain iron-rich materials with Fe2O3 levels above 3%, carbonates below 10%, and a coarse-grained fraction under 25% (Dondi; Raimondo; Zanelli, 2014). Therefore, prior characterization of both clays and raw materials is essential to find necessary adjustments for incorporation into the ceramic mass during the industrial process (Macedo et al., 2008; Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016; Boukili et al., 2021). Additionally, incorporating sludge into bricks can encapsulate pollutants, preventing them from leaching at concentrations exceeding legal limits (Juel; Mizan; Ahmed, 2017).
In this context, the objective of this study was to develop a methodology for predicting the composition of sludge from the white goods metalworking industry (WMI) based on the industrial process, to characterize WMI sludge in terms of pollutants, and to evaluate its potential as a partial substitute for clays used in ceramic brick manufacturing.
2 Materials and methods
2.1 Assessment of pollutants that make up the SMWI of the white goods process
Technical visits were conducted in collaboration with production area professionals to predict the pollutants present in sludge from the metalworking industry (SMWI). This sludge is generated during the physical-chemical treatment of industrial wastewater in the metalworking process of a white goods company in Curitiba.
2.2 Obtaining SMWI and red ceramic production clays
The SMWI samples were provided by a white goods industry in Curitiba. Samples were collected at various stages of the process to capture the variability of SMWI over time. The clays used in this study, named as A0 and B0, were generously donated by ceramic brick manufacturers in Curitiba (Clay A0) and São José dos Pinhais (Clay B0), both in Paraná, Brazil. The modified clays, labeled A5 and B5, were produced by adding 5% dry SMWI (by mass) to the original clays.
2.3 Analysis of SMWI and donated clays
The SMWI, along with the clays provided in their natural state (A0 and B0) and those changed with 5% (by mass, on a dry basis) SMWI (A5 and B5), underwent a series of analyses.
2.3.1 Assessment of the Dangerousness and Inertibility of SMWI
The toxicity of SMWI was evaluated following the methodologies outlined in SW 846 of the United States Environmental Protection Agency (USEPA) Test Methods for Evaluating Solid Waste: Physical/Chemical Methods Compendium - Report Number 846 (USEPA, 1986). Various parameters were assessed using specific methods, including Method 245.7 (1986), Method 3510C (1996), Method 8260C (2006), Method 9010C (2004), Method 9034 (1996), Method 9045D (2004), and Method 9213 (1996). The SMWI was then classified according to its hazardousness and inertness in its natural state, following Brazilian standard NBR 10.004 (ABNT, 2004a), and its leaching extract was evaluated according to NBR 10.005 (ABNT, 2004b) and NBR 10.006 (ABNT, 2004c). A detailed chemical analysis of the solubilized material was performed, focusing on inorganic parameters such as aluminum, arsenic, barium, cadmium, chromium, copper, iron, lead, manganese, mercury, selenium, silver, sodium, zinc, chloride, cyanide, fluoride, nitrate, and sulfate. Additionally, pesticides, including aldrin + dieldrin, chlordane, DDT and its isomers, 2,4-D, endrin, heptachlor and its epoxides, lindane, methoxychlor, pentachlorophenol, toxaphene, 2,4,5-T, and 2,4,5-TP, were identified. The analysis also detected other hazardous organic compounds such as benzene, benzo(a)pyrene, vinyl chloride, chlorobenzene, chloroform, total cresol (m+p), o-cresol, 1,4-dichlorobenzene, 1,2-dichloroethane, 1,1-dichloroethylene, 2,4-dinitrotoluene, hexachlorobenzene, hexachlorobutadiene, hexachloroethane, methyl ethyl ketone, nitrobenzene, pyridine, carbon tetrachloride, tetrachloroethylene, trichloroethylene, 2,4,5-trichlorophenol, and 2,4,6-trichloropheno.
2.3.2 Determination of Loss on Ignition and Thermostability of SMWI and Clays
The loss on ignition (LoI) of the dried (at 105oC to constant mass) samples was found gravimetrically by calcination in a muffle furnace at 1000°C for 2 hours (Parente et al., 2024). Additionally, the mass loss evolution was assessed through thermogravimetric analysis (TGA) using approximately 15 mg of sample. The samples were heated at a rate of 10°C/min from 30°C to 995°C in a nitrogen atmosphere (20 mL/min) using a TGA 4000 System by Perkin Elmer (Bernal et al., 2017).
2.3.3 Determination of SMWI Oxides and Clays
The elements in the SMWI and the clays donated in natura (A0 and B0) were analyzed after being dried in an oven at 105°C for 24 hours. A 7 g part of each dried sample was mixed with a resinous material and pressed for analysis by wavelength-dispersive X-ray fluorescence (WD-XRF) using a PANalytical AXIOS spectrometer equipped with a rhodium X-ray tube (Gazulla Barreda et al., 2016).
2.3.4 Scanning Electron Microscopy and Energy Dispersive Spectrometry Analysis of SMWI and Clays
The residues of the sintered samples from the TGA analysis were examined using scanning electron microscopy (SEM) on a TESCAN VEGA3 LMU microscope at magnifications up to 200kX. Microanalysis of the samples was performed through Energy Dispersive X-ray Spectroscopy (EDS) using an Oxford system equipped with an 80 mm² SDD-type detector. The analysis was conducted on selected fields with the help of AZtech (Advanced) software (Nunes et al., 2023).
3 Results and discussion
This section presents a comprehensive evaluation of the potential use of metalworking sludge (SMWI) as a substitute for clay in ceramic production, offering an alternative to more costly disposal methods such as industrial landfilling. The analysis begins with predicting the potential pollutants present in SMWI, based on the industrial processes and chemicals used in home appliance manufacturing. SMWI was chemically characterized for classification as "solid waste" under Brazilian regulatory frameworks, determining its suitability for reuse or appropriate disposal in line with Brazil's National Solid Waste Policy.
In addition, the chemical characterization of SMWI and two donated clays, commonly conducted in the ceramic industry, enabled comparisons of SMWI with other industrial and wastewater treatment sludges previously proposed as clay additives. The donated clays were also assessed with other reported clay materials.
The study further investigates the feasibility of incorporating up to 5% SMWI into the clays by evaluating the thermostability and microstructural behavior of pure clays and those mixed with SMWI, focusing on their thermal properties and stability. Lastly, the microstructural and compositional analysis of SMWI and clays provides detailed insights into the changes that occur after adding SMWI, supporting the proposed valorization of SMWI to reduce the demand for clay as a raw material in the production of ceramic parts for civil construction.
3.1 SMWI pollutant potentials from the white goods process
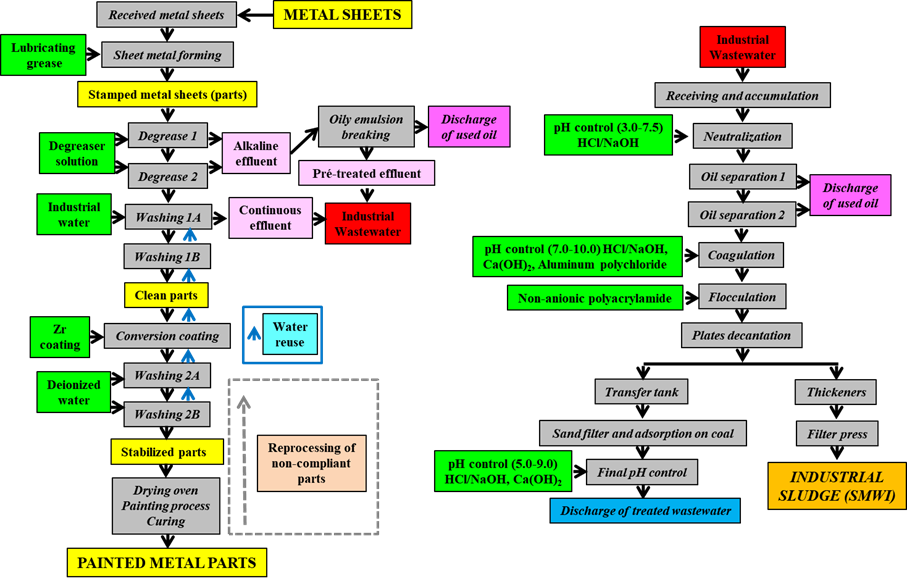

Figure 1: Block diagram of the metal sheet production process and the treatment of Industrial Wastewater (red) that generates sludge (orange) from the metal-mechanical industry (SMWI).
During the production process, metal sheets are initially coated with protective oils and lubricating grease during sheet metal forming. The stamped metal sheets are degreased through an alkaline washing process (Figure 1) using a solution with potassium hydroxide, sodium nitrite, potassium salts, and fatty alcohol (Costa et al., 2012; Costa; Agnoli; Ferreira, 2015). The alkaline effluent generated is pretreated to break emulsions and remove lipid components, which are then sent for specific disposal (e.g., co-processing) and later treated in the industrial wastewater treatment plant (IWT).
Following this, the cleaned metal parts undergo treatment with nanoceramic technology to deposit zirconium layers, enhancing their corrosion resistance (Costa et al., 2012; Costa; Agnoli; Ferreira, 2015; Assemani et al., 2016). These metal parts are washed with deionized water before further operations, without generating more effluent from the process that produces painted metal parts. The alkaline effluent (in light pink, Figure 1) and reused water having dirt, referred to as "continuous effluent" (also in light pink), are combined and sent to the IWT plant (marked in red).
The industrial effluent is then subjected to physical-chemical treatment, which includes pH adjustment with sodium hydroxide or hydrochloric acid for oil separation, coagulation using Ca(OH)2 and aluminum polychloride, and flocculation with anionic polyacrylamide (in green, Figure 1). The solid fraction from this treatment process leads to the formation of SMWI. A critical analysis of this process predicts that SMWI has certain potential components (Table 1), which will be discussed in further detail later in this study.
Table 1: Contaminants predicted in SMWI according to process analysis. |
||
|
Process materials |
Source |
Contaminant |
|
Metal plates |
Metal plate components |
Fe, Cr, Ni, Zn |
|
Defective painted plates (Reprocessing) |
Paint components |
Pigments, organic compounds |
|
Alkaline effluents (Degreasing stages) |
Alkaline treatment |
Oil and greases |
|
Continuous effluents (Washing stages) |
Continuous treatment |
Zr, F, P, Na, CN, phenols |
|
Aluminum polychloride (APC) |
Coagulating agent |
Al, Cl |
|
Anionic polyacrylamide |
Flocculating agent |
Organic compounds |
|
Calcium hydroxide (Lime) |
Pre-alkalizing agent |
Ca, Si |
|
Sodium hydroxide (Soda) |
pH adjustment |
Na |
|
Hydrochloric acid |
pH adjustment |
Cl |
3.2 Chemical analysis of SMWI for classification as solid waste
White goods production process analysis enabled the prediction of the components present in SMWI (Table 1), potentially reducing the need for extensive chemical analyses for its characterization. According to the Brazilian standard NBR 10.004 (ABNT, 2004a), waste is hazardous if it shows flammability, corrosivity, reactivity, toxicity, and/or pathogenicity. Non-hazardous waste, on the other hand, is further classified as inert or non-inert based on the analysis of its solubilized extract (ABNT, 2004a, 2004b, 2004c), which directly affects its disposal options (Sisinno, 2003).
In the case of SMWI, it does not display flammability properties, as the oils are removed during pretreatment. Additionally, the chemicals used in the IWT process, such as hydrochloric acid, polyaluminum chloride, anionic polyacrylamide, calcium hydroxide, and sodium hydroxide, do not have flammability characteristics. SMWI is also not considered corrosive, as its pH stays above 2 and below 12.5 (ABNT, 2004a). It is classified as non-reactive due to its low concentrations of cyanides (<250 mg/kg) and the absence of sulfides (<500 mg/kg) (Table 2). Moreover, SMWI is non-pathogenic, derived solely from the physical-chemical treatment of industrial effluents from metalworking processes, unlike sludge produced by biological treatment in sewage plants (Onofre; Abatti; Tessaro, 2015).
|
Table 2: Characterization of SMWI in natura as set up by Brazilian legal instrument |
|||
|
Classification |
Parameter |
Maximum limit NBR 10.004 (ABNT, 2004) |
Content |
|
“Massa bruta” or in natura |
pH |
2.0 to 12.5 |
8.53 |
|
Sulfide (H2S) |
500 mg/kg |
<4.1 mg/kg |
|
|
Cyanide (HCN) |
250 mg/kg |
0.5 mg/kg |
|
|
Moisture |
% p/p |
75.8% w/w |
|
The mechanical dewatering of SMWI using a filter press reduced its moisture content to 75.8%, which could be further minimized with more efficient equipment or supplementary techniques (Metcalf & Eddy, 2015). This moisture reduction decreases transportation costs and makes it more possible to incorporate SMWI directly into clays for ceramic manufacturing. Toxicity was evaluated through comprehensive chemical analysis using leaching and solubilization tests. In the solubilized fraction, inorganic compounds, pesticides, and other hazardous organic substances were absent, except aluminum, barium, chloride, and total phenols. As a result, SMWI was classified as 'non-inert' (ABNT, 2004a) due to elevated levels of aluminum, chloride, and total phenols in the solubilized material (Table 3), which exceeded the legal limits. Although barium and cresol were detected in the leached fraction, their concentrations remained below the thresholds established by the NBR 10.004 standard (ABNT, 2004a).
|
Table 3: Components detected in the analysis of leached and solubilized extracts from SMWI. |
|||
|
Classification |
Parameter |
Maximum limit (mg/L) NBR 10.004 (ABNT, 2004) |
Content (mg/L) |
|
LEEACH |
|||
|
Inorganic |
Barium |
70.0 |
0.424 |
|
Organic |
Total cresol (m+p) |
200.0 |
0.000472 |
|
SOLUBILIZED |
|||
|
Inorganic |
Chloride |
250.0 |
402* |
|
|
Fluoride |
1.5 |
1.21 |
|
|
Sodium |
200.0 |
96.2 |
|
|
Zinc |
5.0 |
0.279 |
|
|
Aluminum |
0.2 |
0.219 * |
|
|
Barium |
0.7 |
0.132 |
|
|
Copper |
2.0 |
0.0200 |
|
|
Manganese |
0.1 |
0.0110 |
|
Organic |
Total phenols |
0.01 |
0.21* |
Note.: * = it exceeded the limits set by legal standards
In a comparative study of sludge from a similar Brazilian industry, only aluminum (4.45 mg/L solubilized) was found above the legal reference values (Vieira; Silva, 2012), showing potential differences in production and effluent treatment processes. This underscores the importance of individualized analysis of each production process, as the presence or absence of contaminants cannot be generalized.
Specifically, the origin of these pollutants was traced to several sources: fluoride originates from hexafluorozirconate acid used in surface pretreatment processes (Costa; Agnoli; Ferreira, 2015); sodium is introduced via sodium hydroxide used for pH adjustment in IWT; zinc results from the leaching of galvanized metal parts during degreasing with sodium hydroxide; barium, found in the leachate, is derived from pigments in paints used on reprocessed plates (ATSDR, 2016). Further, aluminum and chloride are by-products of polyaluminum chloride, the coagulant used in effluent treatment (Richter, 2001; Martínez-García et al., 2012; Vieira; Silva, 2012, 2012). Phenolic compounds, which are fungicides and bactericides, are traced back to chemical additives applied during metal sheet treatment and painting processes (ATSDR, 2016), with cresol residues (1-hydroxy-4-methylbenzene) originating from preservatives or oils removed from the sheets.
Most components of SMWI can be associated to the materials used in the process (Table 1), except for silica (SiO2), which is a contaminant from lime. Based on these findings, SMWI is classified as a "Non-Hazardous Waste Class II A – Non-Inert" (ABNT, 2004a). These results confirm the efficiency of process analysis as a method to reduce the need for extensive chemical testing, emphasizing the importance of thoroughly understanding all unit operations and chemical components involved.
Historically, SMWI has been disposed of in industrial landfills, and more recently, has been used in cement co-processing and as a binding agent in civil construction, with the latter options offering lower costs. Industrial landfill disposal is particularly costly and environmentally detrimental, making it a less desirable choice (Sisinno, 2003). Additionally, energy recovery from SMWI is limited by chlorine (Table 3), as outlined by the parameters in CONAMA Resolution No. 499 (BRASIL, 2020).
Based on Brazil’s waste disposal hierarchy (BRASIL, 2010), the recovery of coagulants from sludge (Metcalf & Eddy, 2015; Ahmad; Ahmad; Alam, 2016) was considered economically unfeasible due to the complexity of the required operations. Therefore, integrating sludge into raw materials for ceramic production (Vieira; Silva, 2012; Tartari et al., 2011; Sutcu et al., 2022; Coletti et al., 2016; Juel; Mizan; Ahmed, 2017; Hasan; Hashem; Payel, 2022; Munir et al., 2018; Simón et al., 2021; Benlalla et al., 2015; Oliveira; Holanda, 2008; Martínez-García et al., 2012; Zhang et al., 2016; Ukwatta et al., 2015; Ukwatta et al., 2016; Ukwatta; Mohajerani, 2017) emerges as a viable alternative, provided it does not compromise the quality of the construction products. This approach not only renders SMWI inert but also reduces the need for primary raw materials, thereby offering significant environmental and human health benefits associated with their extraction and processing (Tartari et al., 2011; Sutcu et al., 2022).
3.3 Chemical analyses of SMWI and donated clays
3.3.1. Moisture determination of SMWI in natura and donated clays
The moisture content of SMWI (75.8%) was more than double that of the donated clays (A0 = 29.0% and B0 = 24.0%). This is a crucial factor in the preparation of the ceramic mass before the molding process, whether through extrusion or dry pressing (Dondi; Raimondo; Zanelli, 2014). The high moisture content of SMWI also raises concerns about transportation planning, as the excess water increases the need for more vehicles, fuel, personnel, and associated costs.
3.3.2. Determination of loss on ignition of SMWI and donated clays
"Loss on ignition" (LoI) refers to the weight loss of a material when subjected to elevated temperatures, used to find the content of volatile substances such as crystalline water, organic matter, carbonates, and other compounds that decompose or evaporate at elevated temperatures. For instance, the dehydroxylation of kaolinite (Al₂O₃·2SiO₂·2H₂O) occurs between 400°C and 650°C (Ptáček et al., 2014). The LoI of the unadditivated donated clays (A0 = 10.28% and B0 = 11.37%) was consistent with the average reported for other clays (avg. = 10.63%, standard deviation or DP = 3.76%, and range or R = 14.31%). However, this value was only about 25% of the LoI for the dry SMWI (39.53%), where the LoI can be primarily attributed to organic matter, carbonates, and other materials that are driven off from the sample (Teixeira et al., 2011; Martínez-García et al., 2012; Zhang et al., 2016; Yara, 2020).
3.3.3 Determination of oxides in donated clays
The composition of clays used in commercial ceramics production is primarily composed of SiO₂, Al₂O₃, Fe₂O₃, and CaO (Figure 2), which together account for an average of 90.38% (with a DP of 4.70% and an R of 15.04%) of the total oxide content. This composition can vary widely depending on the clay's extraction area (Macedo et al., 2008) and the specific clay minerals present (Dondi; Raimondo; Zanelli, 2014). SiO₂ and Al₂O₃ are the predominant oxides in these clays, essential for the formation of common clay minerals like kaolinite, illite, montmorillonite, and chlorite, which often contain significant amounts of SiO₂ and Al₂O₃ within their crystalline structures (Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016).
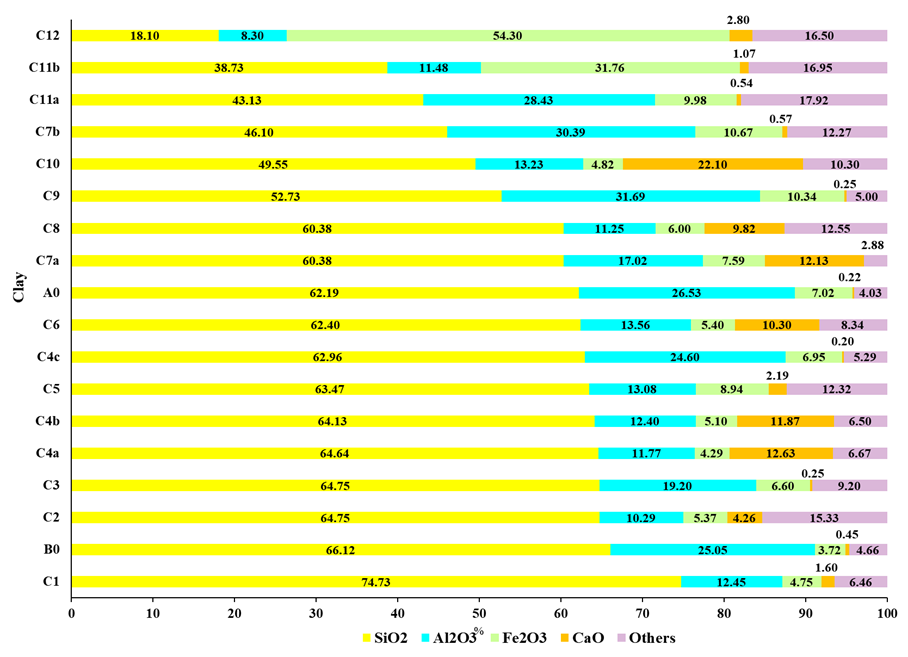
Figure 2: Main oxides in clays.
Note.: C1 (Yaras, 2020), B0 (clay donated from São José dos Pinhais), C2 (Hasan; Hashem; Payel, 2022), C3 (Ukwatta; Mohajerani, 2017), C4a, C4b e C4c (Boukili et al., 2021), C5 (Juel; Mizan; Ahmed, 2017), C6 (Martínez-García et al., 2012), A0 (clay donated from Curitiba), C7a e C7b (Benlalla et al., 2015), C8 (Munir et al., 2018), C9 (Oliveira; Holanda, 2008), C10 (Coletti et al., 2016), C11a e C11b (Tartari et al., 2011); C12 (Simón et al., 2021).
The analysis of donated clays (A0 and B0) alongside reported clays show a lack of linear correlation between the major oxides (SiO₂, Al₂O₃, Fe₂O₃, and CaO), which highlights the natural variability in clay composition. SiO₂ content typically exceeds 45%, as seen in both donated clays (A0 = 62.19%, B0 = 66.12%) and most of the reported clays (avg. = 56.63%, SD = 12.94, R = 56.63%), although there are exceptions, such as C12, with only 18.10% SiO₂ (Figure 2). Al₂O₃ is the second most abundant component, confirmed in the donated clays (A0 = 26.53%, B0 = 25.05%) and most reported samples (avg. = 16.82%, SD = 7.55%, R = 23.39%), with some anomalies like C11b and C12.
Fe₂O₃ content, while not always significant, is typically above 3% in clays used for red ceramics (Dondi; Raimondo; Zanelli, 2014), and this trend is reflected in the donated clays (A0 = 7.04%, B0 = 3.72%) and most reports (avg. = 11.43%, SD = 12.18%, R = 50.01%), although with outliers such as C11b (31.76%) and C12 (54.30%). The CaO content is typically low, as seen in the donated clays (A0 = 0.22%, B0 = 0.45%) and in certain samples such as C4c, C3, C9, C11a, and C7b. Other oxides are 4.03% of A0 and 4.66% of B0, with broader variability in their content across different samples (avg. = 17.92%, SD = 2.88%, R = 15.04%).
The wide variation in clay oxides used in red ceramics production (Macedo et al., 2008; Dondi; Raimondo; Zanelli, 2014) presents a significant opportunity for incorporating various solid wastes, including SMWI (Sludge from Metalworking Industry). The clays A0 and B0, which are rich in specific oxides, could potentially incorporate high SMWI content, making them an environmentally desirable destination for this waste.
However, it is essential to consider the functional roles of these oxides in the resulting ceramics. For instance: a) SiO₂: Reduces plasticity and linear shrinkage while increasing ceramic porosity. b) Al₂O₃: Enhances mechanical strength (Muñoz Velasco et al., 2014). c) Fe₂O₃: In concentrations above 3-4%, it generates red ceramics upon firing (Ukwatta; Mohajerani, 2017; Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016; Rodrigues; Della Sagrillo; Reis, 2020). If Fe₂O₃ is below 1-2%, it results in white ceramics (Dondi; Raimondo; Zanelli, 2014).
The clays' composition also includes other elements (e.g., K₂O, TiO₂, MgO, Na₂O, P₂O₅, SO₃, ZrO₂, BaO, and MnO in A0) that contribute to the desired properties of ceramics, such as high mechanical strength (Dondi; Raimondo; Zanelli, 2014; Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016).
Wavelength-dispersive X-ray fluorescence (WD-XRF) spectrometry allows for precise and accurate quantification of both major and trace elements, such as Ba, Ce, Co, Cr, Cu, Fe, La, Mn, Ni, Pb, Rb, S, Sr, Ta, Th, U, V, Y, Zn, and Zr (Gazulla Barreda et al., 2016). This analysis is critical in deciding the suitability of the clays for ceramic production, especially when incorporating SMWI, as it ensures that the final product keeps the desired mechanical and aesthetic properties. In conclusion, the incorporation of SMWI into clays A0 and B0 is promising, but careful consideration of oxide content (Table 4) and its effects on the final ceramic properties is crucial for achieving best results.
|
Table 4: Chemical composition of SMWI and clays by WD-XRF
|
||||||||||||||
|
Sample |
SiO2 |
Al2O3 |
Fe2O3 |
CaO |
K2O |
TiO2 |
MgO |
Na2O |
P2O5 |
ZnO |
Cl |
SO3 |
SrO |
|
|
SMWI |
7.28 |
25.81 |
0.66 |
43.02 |
3.81 |
0.17 |
2.48 |
1.82 |
6.62 |
4.47 |
3.15 |
0.33 |
0.17 |
|
|
A0 |
62.19 |
26.53 |
7.02 |
0.22 |
1.23 |
1.56 |
0.56 |
0.11 |
0.11 |
ND |
ND |
0.11 |
ND |
|
|
B0 |
66.12 |
25.05 |
3.72 |
0.45 |
1.58 |
1.69 |
0.56 |
0.23 |
0.11 |
ND |
ND |
0.11 |
ND |
|
Note.: ZrO2 (0.11%), BaO (0.11%) and MnO (0.11%) were found in A0. ZrO2 (0.11%) and BaO (0.11%) were found in B0. ND = Not Detected.
3.3.4 Determination of SMWI oxides and association with the source of the industrial process
The analysis of SMWI (Sludge from the Metalworking Industry) revealed that its composition is primarily influenced by the chemicals used during effluent treatment and surface pretreatment of metal parts. Specifically, the major part, CaO (43.02%), originates from the use of Ca(OH)2 as a coagulation adjuvant in the industrial effluent treatment process (Figure 1, Table 1). The high content of Al₂O₃ (25.81%) is attributed to the use of polyaluminum chloride as a coagulating agent, which also contributes to the presence of Cl (3.15%), as Cl can also be traced back to the use of hydrochloric acid for pH control.
Na₂O (1.82%) is due to the use of sodium hydroxide in pH adjustment. It is important to note that part of the sodium chloride and other thermolabile compounds might volatilize during the loss on ignition (LoI), alongside the decomposition of precipitated salts or the dehydroxylation of crystal structures (Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016; Rodrigues; Della Sagrillo; Reis, 2020). ZnO (4.47%) and Fe₂O₃ (0.66%) originated from the washing of metal parts that undergo leaching during surface pretreatment. Other oxides present in SMWI, such as SiO₂ (7.28%), P₂O₅ (6.62%), K₂O (3.81%), MgO (2.48%), and TiO₂ (0.17%), could originate from various stages of the surface pretreatment process, additives in the degreasing solutions, metal surface treatments, or even from contaminants introduced during these processes.
In summary, the composition of SMWI directly reflects the chemicals used in the metalworking process and effluent treatment, with major oxides like CaO and Al₂O₃ contributing playing crucial roles due to their respective uses in coagulation and pH control. The presence of other oxides underscores the complexity of the sludge composition and its potential impact on later processing or disposal options.
3.4 Comparison between the composition of SMWI and other sludges
Due to the acceptable variation in the oxide composition of clays used in the production of red ceramics (Figure 2), various sludges (Figure 3) from different sources—including sewage treatment plant (2 S-STP, 3 S-STP, 4 S-STP, and 5 S-STP), water treatment plant (6 S-WTP, 7 S- WTP, 8 S- WTP, and 9 S- WTP), and several industrial processes such as ceramics (1 S-CER), paper milling (10 S-PAP), tanning (11 S-TAN and 15 S-TAN), dairy processing (12 S-DAI), marble processing (13 S-MAR), carbonation (14 S-CO2), and metalworking (16 S-MMI), which is the focus of this study (SMWI)—can be considered potential raw materials for ceramic production. These sludges have between 56.55% and 99.39% of a mixture of SiO2, Al2O3, Fe2O3, and CaO. This finding also suggests other, less obvious opportunities, such as the disposal of sargassum stranded on beaches, which is organic. Although life cycle analysis has shown that the sintering of algae into ash may not be incredibly helpful, it presents a workable option for addressing beach maintenance challenges, offering a potentially definitive solution with social, economic, and environmental benefits (Parente et al., 2024).
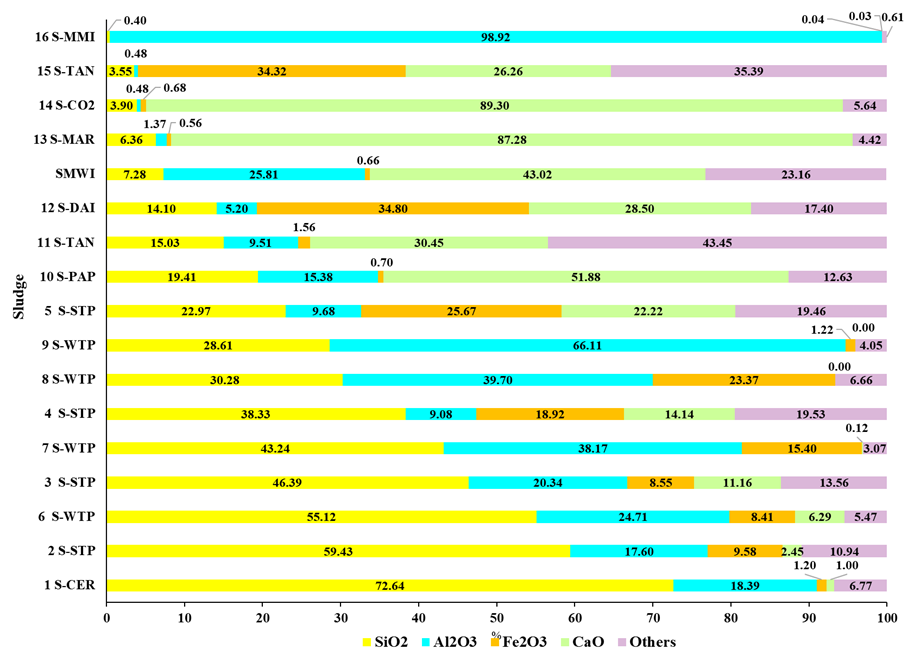
Figure 3: Main oxides in sludge
Note.: 1 S-Cer= Ceramic (Coletti et al., 2016); 2 S-STP= Sewage Treatment Plant (Ukwatta et al. 2016); 6 S-WTP= Water Treatment Plant (Sutcu et al., 2022); 3 S-STP (Martínez-García et al., 2012); 7 S-WTP (Oliveira; Holanda, 2008); 4 S-STP (Ukwatta; Mohajerani, 2017); 8 S-WTP (Tartari et al., 2011); 9 S-WTP (Benlalla et al., 2015); 5 S-STP (Zhang et al., 2016); 10 S-PAP= Paper milling (Yaras, 2020); 11 S-TAN= Tannery (Hasan; Hashem; Payel, 2022); 12 S-DAI= Dairy (Simón et al., 2021); SMWI; 13 S-MAR= Marble (Munir et al., 2018); 14 S-CO2= Carbonation (Yaras, 2020); 15 S-TAN= Tannery (Juel; Mizan; Ahmed, 2017); 16 S-MMI= Metalworking (Vieira; Silva, 2012)
Silicon dioxide (SiO2) is a desirable part in clays used for ceramic production (Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016). Consequently, the sludges analyzed in this study were coded according to their SiO2 content in decreasing order, except for those from sewage treatment plant (STP) and water treatment plant (WTP). The sludge from the ceramic industry (1 S-CER) showed the highest SiO2 content at 72.64%, which is consistent with its industrial origin. SiO2 levels in STP and WTP sludges ranged from 59.43% to 22.97% and 55.12% to 28.61%, respectively, aligning with the presence of sand residues and contaminants from coagulation reagents used in water treatment plant (Richter, 2001; Metcalf & Eddy, 2015). Despite the wide variability in SiO2 content across industrial sludges (0.40%-72.65%), their incorporation into ceramic production is still possible, albeit with limited addition.
The highest SiO2 content was seen in ceramic industry sludge (1 S-CER), followed by STP sludge and WTP sludge. In STP, SiO2 primarily originates from sand particles, while in WTP, it is derived from contaminated CaO used during coagulation (Richter, 2001; Metcalf & Eddy, 2015). Industrial sludges generally contain lower SiO2 levels (0.40%-19.41%), reflecting the raw materials and sewage treatment processes employed. The SMWI (sludge from metalworking industry) presents intermediate SiO2 levels (7.28%), which are significantly lower compared to the lowest SiO2 content in clay (18.10% in C12), making direct use of SMWI as a raw material for ceramic production unfeasible. Al2O3 content varied widely from 0.48% (14 S-CO2 and 15 S-TAN) to 98.92% (16 S-MMI), with combined SiO2 and Al2O3 contents ranging from 4.03% to 99.32%. These variations need cautious addition to ensure the formation of minerals that contribute desirable mechanical properties to the ceramics (Macedo et al., 2008; Dondi; Raimondo; Zanelli, 2014).
The Fe2O3 content was notably higher in sludges from dairy (12 S-DAI), tanning (11 S-TAN), and certain STP and WTP treatments, suggesting the possible use of iron ions as a coagulation agent (Richter, 2001; Metcalf & Eddy, 2015). These sludges could serve as additives in red ceramics, while others could be used in white ceramics (Dondi, Raimondo, Zanelli, 2014). SMWI, with a higher Al2O3 content (25.81%) and lower Fe2O3 content (0.66%), appears to be a superior additive compared to most other sludges, except for the sludge from another metalworking industry (16 S-MMI), which has 98.92% Al2O3 and 0.04% Fe2O3. However, it is important to note that sludge from the metalworking industry is not universally the best additive, as this depends on the specific effluent treatment processes applied.
Calcium oxide (CaO) is present in over 25% of industrial sludges (14 S-CO2, 13 S-MAR, 10 S-PAP, SMWI, 11 S-TAN, 12 S-DAI, 15 S-TAN), except for 1 S-CER and 16 S-MMI, which can be attributed to the raw material matrix and effluent treatment principles. The metalworking sludge from another Brazilian industry (16 S-MMI) was composed of Al2O3 (98.92%), with small amounts of other oxides such as SO3, SiO2, K2O, Fe2O3, CaO, Cr2O3, ZnO, MnO, and CuO (Vieira, Silva, 2012). The differences between this sludge and SMWI can be attributed to variations in the treatment processes and the operational diversity of industrial effluent treatment plants.
In conclusion, the similar Al2O3 content between SMWI and clays (A0 and B0) suggests that their mixing is possible (Table 4). However, this is not the case for SiO2. Nonetheless, the addition of SMWI can be improved based on the composition of other clays like C18b (SiO2=46.10%, Al2O3=30.39%) and C8b (SiO2=43.13%, Al2O3=28.43%). The high CaO content (43.0%) in SMWI can compensate for its low levels in A0 (0.22%) and B0 (0.45%), potentially enhancing the mechanical strength of the final ceramic product by reacting with SiO2 in the clay. However, the addition must be carefully controlled to prevent the formation of free CaO in ceramics used in civil construction, which could lead to undesirable water absorption (Muñoz Velasco et al., 2014; Rodrigues; Della Sagrillo; Reis, 2020). The presence of other oxides (23.16%), such as K2O (3.81%), MgO (2.48%), and Na2O (1.82%), contributes to the formation of minerals like illite, montmorillonite, and chlorite, which enhance the mechanical strength of the ceramics (Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016).
Therefore, SMWI can be considered a desirable additive for both red and white ceramic industries (Muñoz Velasco et al., 2014; Rodrigues; Della Sagrillo; Reis, 2020), ending the costs associated with its disposal in industrial landfills or stabilization treatment. The valorization of this waste reduces the overall production costs for construction materials (Sutcu et al., 2022) and has a socially beneficial effect by minimizing competition for land use (Parente et al., 2024). Further studies are needed to decide the technical feasibility of using SMWI as an additive in red ceramic production.
3.5 Thermostability of SMWI, pure clays and with 5% of their addition
The mass evolution during the sintering process, as predicted by thermogravimetric analysis (TGA) during brick firing, shows similar profiles for the donated clays (A0 and B0) and the clays with 5% additive (by mass, A5 and B5), with some variation in the peak temperatures for specific events (Figure 4a and 4b). The mass loss (WL) of the untreated SMWI was 83.17% between 30°C and 995°C. Specifically, for SMWI, the mass loss (WLSMWI) was 40.96% up to 105°C and 71.40% up to 155°C (Figure 4c), with peaks at 93°C and 103°C, which can be attributed to the elimination of free and adsorbed or occluded water (Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016; Boukili et al., 2021). The WLSMWI up to 600°C was 6.23% (Table 5), consistent with the decomposition of organic matter (Yara, 2020; Zhang et al., 2016), hydroxides (Teixeira et al., 2011), and the elimination of low-volatility compounds (Martínez-García et al., 2012). The lower WLSMWI between 155°C and 600°C compared to the clays aligns with the lower organic compound content in industrial waste and the presence of high levels of Al2O3 and CaO, which contribute to the formation of thermostable clay minerals (Savazzini-Reis; Della-Sagrillo; Valenzuela-Diaz, 2016).
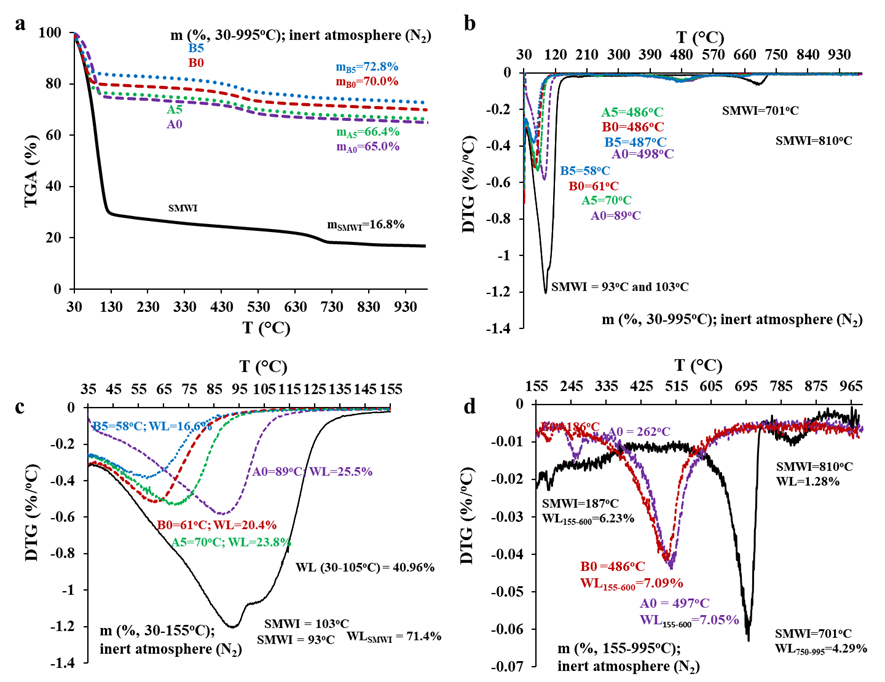
Figure 4: Evolution of mass losses (WL) by TGA of SMWI and donated clays (A0 and B0) and additives at 5% with SMWI (A5 and B5) (a) and derivative curves for temperatures between 30oC and 995oC (b), 30oC and 155oC (c) and 155oC and 995oC (d).
|
Table 5: Evolution of mass loss (%) of SMWI and clays by TGA
|
|
||||||
|
Sample |
30-155ºC |
155-600ºC |
600-750ºC |
750-950ºC |
950-990ºC |
30-995ºC |
Residual |
|
SMWI |
71.36 |
6.23 |
4.29 |
1.15 |
0.13 |
83.17 |
16.83 |
|
A0 |
25.48 |
7.11 |
1.04 |
1.10 |
0.29 |
35.04 |
64.96 |
|
B0 |
20.43 |
7.09 |
1.02 |
1.23 |
0.26 |
30.02 |
69.98 |
|
A5 |
23.79 |
7.05 |
1.39 |
1.17 |
0.22 |
33.62 |
66.38 |
|
B5 |
16.59 |
7.52 |
1.59 |
1.25 |
0.26 |
27.20 |
72.80 |
Note: SMWI samples A0 and B0 were in natura. A5 and B5 received dry SMWI to make up 5% (on a dry basis).
Notably, Al2O3 can react with silica at vitrification temperatures (1000-1650ºC), forming mullite (1.4Al2O3.SiO2) (Lima et al., 2022), which enhances the mechanical strength of ceramic materials (Muñoz Velasco et al., 2014; Ukwatta; Mohajerani, 2017), thereby offering new opportunities for incorporating SMWI into other ceramic products. Similarly, CaO can contribute to increased mechanical strength if it forms bonds with SiO2 (Muñoz Velasco et al., 2014; Rodrigues; Della Sagrillo; Reis, 2020). Additionally, the weight loss (WL) of 75.8%, corresponding to the moisture content in the characterization of the untreated SMWI (Table 2), was seen up to 450°C. However, this result should be interpreted with caution due to differences in analytical techniques and the potential volatilization of substances other than water (Bernal et al., 2017).
Peaks at 701°C and 810°C (Figure 4d) are attributed to the decarbonation of calcium carbonate (Munir et al., 2018; Yaras, 2020; Martínez-García et al., 2012; Boukili et al., 2021) and the formation of illite ((K,H3O)(Al,Mg,Fe)2(Si,Al)4O10[(OH)2,(H2O)]) using various oxides from SMWI (Findik; Akyol; Sari, 2014). Finally, the WLSMWI between 105°C and 995°C (42.21%) was slightly higher than the loss on ignition (LoI) value of 39.53%, due to the inherent differences between the techniques and the varying atmospheres—oxidizing in LoI and inert in TGA (Bernal et al., 2017).
The WL of A0 up to 155°C was higher than that of B0, which are both ready-to-use clays, showing distinct properties such as differences in organic matter content and water retention capacity (Dondi; Raimondo; Zanelli, 2014). The WL for additive clays (A5 and B5) was slightly lower (Table 5) than for the donated clays (A0 and B0), possibly due to the addition of dry SMWI. The first peak for B0 occurred earlier (61ºC) than for A0 (89ºC) (Figure 4c), and the additive forms shown even earlier peaks (WLB5=58ºC and WLA5=70ºC) compared to their matrices (A0 and B0). The WL between 155ºC and 600ºC was similar for A0 and B0, slightly higher than for A5 and B5, consistent with the lower WLSMWI. Peaks occurring between 450°C and 600°C (Figure 4d) in the donated and additive clays are associated with the dehydroxylation of clay minerals (loss of structural water), such as kaolinite (Al2O3·2 SiO2·2H2O) dehydroxylation between 400°C and 650°C (Ptáček et al., 2014), and the decomposition of organic matter (Ukwatta et al., 2015; Zhang et al., 2016).
In this context, the thermostability of the donated clays mixed with 5% SMWI (A5 and B5) aligns with the formation of thermostable clay minerals in ceramics, highlighting the potential of SMWI as a beneficial additive in ceramic materials.
3.6 Microstructural and micro-composition analysis of SMWI and clays
The microstructure of the SMWI sintered in TGA (Figure 5a) showed a surface characterized by larger cracks and pores compared to those saw in A0 and B0. This appearance is expected due to the substantial amount of water randomly distributed among the particles, which is removed during sample preparation. In contrast, the relatively uniform appearance of A0 and B0 (Figures 5b and 5c) can be attributed to their prior preparation for red ceramic production, resulting in more homogeneous masses.
|
(a) |
(b) |
(c) |
||||||
|
Figure 5: Images of SMWI (a), A0 clay (b), and B0 clay (c) by SEM.
|
||||||||
Energy-dispersive X-ray spectroscopy (EDS) analysis of the SMWI (Table 6) revealed elevated levels of oxygen (23.4%) and carbon (36.7%), due to the presence of oxides and carbonates, as reported by Vieira and Silva (2012). The carbon content in SMWI was comparable to that of A0, while the oxygen content was like that of both B0 and SMWI. When comparing the composition using oxide equivalents, Al2O3 and CaO are predominant in SMWI. However, discrepancies between analysis methods were noted (Table 7), particularly when comparing results from EDS and wavelength-dispersive X-ray fluorescence (WD-XRF). For instance, CaO content was measured at 43.04% by WD-XRF, but 51.70% by EDS, a 20% increase in the latter. Similarly, discrepancies were seen for other elements, with EDS showing higher values for Fe2O3 and SiO2 and lower values for Al2O3 and K2O. The most significant discrepancy was for TiO2, where the EDS value was 207% higher.
Table 6: Average (n=5) proximate chemical composition of SMWI and clays by EDS during SEM
|
Sample |
Si |
Al |
Fe |
Ca |
K |
Ti |
Mg |
Na |
P |
C |
O |
|
SMWI |
2.20 |
7.80 |
0.60 |
23.60 |
0.30 |
0.20 |
1.30 |
0.30 |
1.90 |
23.4 |
36.7 |
|
A0 |
16.00 |
11.10 |
4.50 |
0.20 |
0.80 |
0.60 |
0.30 |
ND |
ND |
24.0 |
42.5 |
|
B0 |
27.10 |
9.40 |
4.70 |
0.10 |
0.60 |
0.50 |
0.20 |
ND |
ND |
18.8 |
38.4 |
|
|
SiO2 |
Al2O3 |
Fe2O3 |
CaO |
K2O |
TiO2 |
MgO |
Na2O |
P2O5 |
|
|
|
SMWI |
8.78 |
23.07 |
1.34 |
0.57 |
0.52 |
3.38 |
0.63 |
6.82 |
0.00 |
0.00 |
|
|
A0 |
53.17 |
32.58 |
9.99 |
0.43 |
1.50 |
1.56 |
0.77 |
ND |
ND |
0.00 |
0.00 |
|
B0 |
68.49 |
20.98 |
7.94 |
0.17 |
0.85 |
0.99 |
0.39 |
ND |
ND |
0.00 |
0.00 |
Note: ND = not detected. The conversion of elemental values into their respective oxides was carried out using the atomic weight of the ChemicalAid (https://www.chemicalaid.com/) and the O and C values were purged to make up 100% of matter.
|
Table 7: Relationship between the average approximate chemical composition (n=5) of SMWI and clays by EDS during SEM and the chemical composition of SMWI and clays by WD-XRF |
|||||||||
|
Sample |
SiO2 |
Al2O3 |
CaO |
K2O |
TiO2 |
MgO |
Na2O |
P2O5 |
|
|
SMWI |
1.21 |
0.89 |
2.03 |
1.20 |
0.15 |
3.07 |
1.36 |
0.35 |
1.03 |
|
A0 |
0.85 |
1.23 |
1.42 |
1.98 |
1.22 |
1.00 |
1.38 |
ND |
ND |
|
B0 |
1.04 |
0.84 |
2.13 |
0.37 |
0.54 |
0.58 |
0.70 |
ND |
ND |
Note: ND = not detected.
These discrepancies are also clear in the analysis of A0 and B0 (Table 7), likely due to the heterogeneity of the samples analyzed by EDS. The variation in the analyzed fields becomes clearer when the analyzed points are magnified, revealing particles that are the overall sample composition (Table 8). It is important to note that these clays, despite being considered suitable for industrial use, are still fragmentary and heterogeneous. For example, the Ti content in A0 ranged from 0.4% to 2.1%, and in B0, it ranged from 0.4% to 15.4%. While this variation is less pronounced for major elements, it is significant enough to affect the reliability of the analysis under the conditions used.
|
Table 8: Spot chemical composition of clays by EDS during SEM |
||||||||||
|
Sample |
Point |
O |
C |
Si |
Al |
Fe |
Ca |
K |
Ti |
Mg |
|
A0 |
P1 |
35.9 |
36.1 |
9.6 |
9.3 |
5.4 |
0.3 |
0.5 |
1.2 |
0.2 |
|
P2 |
36.7 |
31.2 |
13.2 |
9.6 |
7.3 |
0.1 |
0.8 |
0.8 |
0.2 |
|
|
P3 |
20.4 |
26.3 |
8.9 |
7.0 |
36.5 |
0.2 |
0.4 |
0.4 |
0.2 |
|
|
P4 |
45.0 |
15.0 |
18.4 |
12.3 |
6.1 |
0.1 |
0.6 |
2.1 |
0.3 |
|
|
P5 |
44.2 |
12.7 |
14.4 |
11.8 |
15.6 |
0.2 |
0.5 |
0.6 |
ND |
|
|
P6 |
38.4 |
18.8 |
27.1 |
9.4 |
4.7 |
0.1 |
0.6 |
0.5 |
0.5 |
|
|
B0 |
P1 |
43.5 |
6.5 |
11.7 |
7.8 |
13.8 |
0.2 |
0.3 |
15.4 |
0.3 |
|
P2 |
49.4 |
9.2 |
24.4 |
12.1 |
1.8 |
0.2 |
1.8 |
0.4 |
0.3 |
|
|
P3 |
43.3 |
10.8 |
18.6 |
11.0 |
8.5 |
0.2 |
3.0 |
1.4 |
2.8 |
|
|
P4 |
47.3 |
8.7 |
24.8 |
14.5 |
2.6 |
0.4 |
0.7 |
0.6 |
0.4 |
|
|
P5 |
48.5 |
12.4 |
19.6 |
14.7 |
2.4 |
ND |
0.9 |
0.6 |
0.4 |
|
|
P6 |
47.3 |
11.3 |
22.6 |
14.3 |
2.4 |
0.4 |
0.6 |
0.6 |
0.6 |
|
Note: ND = not detected.
To improve precision and reliability, samples should be ground into exceptionally fine particles (< 2 µm) to complement X-ray diffraction (XRD) results (Clayton; Pearce, 2007). In this context, WD-XRF results should be considered more reflective of the actual composition of SMWI, A0, and B0 (Gazulla Barreda et al., 2016). It is also important to note that while WD-XRF provides elemental data on the chemical composition without differentiating between specific chemical compounds, XRD names and quantifies the minerals and their phases, offering a more comprehensive analysis of the sample’s composition and structure (QUANTUM ANALYTICS, 2024).
4 Conclusions
This study evaluated the potential of using metallurgical sludge from the metalworking industry (SMWI) as an additive in clays used for brick production. For this purpose, SMWI and two industrial clays prepared for brick manufacturing were characterized thermally, chemically, and microstructurally. This innovative study conducted a detailed analysis of the generation, treatment, and composition of SMWI, proving its viability as an additive in ceramic production. Quantitative chemical analysis confirmed that SMWI is predominantly inorganic, containing components such as Si, P, Zn, K, Fe, Mg, Ti, Na, Cl, Al, and Ca, derived from the pretreatment and surface washing of metal parts and the reagents used in the Effluent Treatment Plant (ETP). Although SMWI does not exactly match the composition of sewage and industrial sludges typically used in ceramics, its composition rich in SiO2, Al2O3, Fe2O3, and CaO makes it suitable to produce both red and white ceramics. The high CaO content can enhance the mechanical strength of ceramics, though excess CaO could reduce strength and increase water absorption. The SiO2 in SMWI mitigates these effects, but its addition should be limited to around 5% by mass. Utilizing SMWI as a ceramic raw material enables the immobilization of pollutants within the ceramic matrix or their thermal degradation during manufacturing. For efficient production, SMWI must be adequately fragmented and mixed, ensuring a homogeneous mass despite the anisotropy of the particles. Therefore, substituting clay with SMWI in brick manufacturing presents a promising alternative for waste valorization and reducing the environmental impact associated with clay extraction. Further studies are recommended to evaluate the physical and mechanical properties of ceramics produced with this residue to ensure their technical feasibility according to Brazilian standards.
5 Acknowledgements
We thank CAPES, CNPq, DAAD, and UFPR for their invaluable financial and structural support in this research endeavor.
Bibliographic references
ABNT. ASSOCIAÇÃO BRASILEIRA DE NORMAS TÉCNICAS. NBR 10.005: Procedimento para obtenção de extrato lixiviado de resíduos sólido. Rio de Janeiro, (2004b). Disponível em: https://www.normas.com.br/visualizar/abnt-nbr-nm/4033/nbr10005-procedimento-para-obtencao-de-extrato-lixiviado-de-residuos-solido. Acesso em: 22 mai. 2024.
ABNT. ASSOCIAÇÃO BRASILEIRA DE NORMAS TÉCNICAS. NBR 10.006: Procedimento para obtenção de extrato solubilizado de resíduos sólidos. Rio de Janeiro, (2004c). Disponível em: https://www.normas.com.br/visualizar/abnt-nbr-nm/6013/nbr10006-procedimento-para-obtencao-de-extrato-solubilizado-de-residuos-solidos. Acesso em: 22 mai. 2024.
AHMAD, T.; AHMAD, K.; ALAM, M. Sustainable management of water treatment sludge through 3’R’ concept. Journal of Cleaner Production, v. 124, 1-13, 2016. https://doi.org/10.1016/j.jclepro.2016.02.073. Acesso em: 22 mai. 2022. Disponível em: https://is.gd/MQqYeA. Acesso em: 22 mai. 2024.
ASSEMANI, H. R.; AHMADI, P.; SARABI, A. A.; EIVAZ MOHAMMADLOO, H. Effect of zirconium conversion coating: Adhesion and anti-corrosion properties of epoxy organic coating containing zinc aluminum polyphosphate (ZAPP) pigment on carbon mild steel. Progress in Organic Coatings, v. 94, 18–27, 2016. https://doi.org/10.1016/j.porgcoat.2016.01.015. Disponível em: https://is.gd/vnhlgx. Acesso em: 22 mai. 2024.
ATSDR. AGENCIA PARA SUSTANCIAS TOXICAS Y EL REGISTRO DE ENFERMEDADES, ToxFAQsTM - Índices Alfabéticos. 2016. Disponível em: https://is.gd/HfxNns. Acesso em: 22 mai. 2024.
BENLALLA, A.; ELMOUSSAOUITI, M.; DAHHOU, M.; ASSAFI, M. Utilization of water treatment plant sludge in structural ceramics bricks. Applied Clay Science. v. 118, 171–177, 2015. https://doi.org/10.1016/j.clay.2015.09.012. Disponível em: https://is.gd/czr8TW. Acesso em: 22 mai. 2024.
BERNAL, S. A.; JUENGER, M. C. G.; KE, X.; MATTHES W.; LOTHENBACH, B.; DE BELLE, N.; PROVIS, J. L. Characterization of supplementary cementitious materials by thermal analysis. Materials and Structures, v.50, n.26, 1-13, 2017. DOI 10.1617/s11527-016-0909-2. Disponível em: https://is.gd/WfC8Vl. Acesso em: 22 mai. 2024
BOUKILI, G. E.; LECHHEB, M.; OUAKARROUCH, M.; DEKAYIR, A; KIFANI-SAHBAN, F.; KHALDOUN, A. Mineralogical, physico-chemical and technological characterization of clay from Bensmim (Morocco): Suitability for building application. Construction and Building Materials, v. 280, 122300, 2021. https://doi.org/10.1016/j.conbuildmat.2021.122300. Disponível em: https://is.gd/h9oDU9. Acesso em: 22 mai. 2024.
BRASIL. Lei n° 12.305 de 2 de agosto de 2010. Institui a Política Nacional de Resíduos Sólidos; altera a Lei no 9.605, de 12 de fevereiro de 1998; e dá outras providências. Diário Oficial da União, Brasília, DF, n. 227, 02 ago. 2010. Disponível em: https://is.gd/GJ5rlw. Acesso em: 22 mai. 2024.
BRASIL. Resolução CONAMA n° 430, de 13 de maio de 2011. Dispõe sobre as condições e padrões de lançamento de efluentes, complementa e altera a Resolução no 357, de 17 de março de 2005, do Conselho Nacional do Meio Ambiente-CONAMA. Disponível em: https://is.gd/2IBiLh. Acesso em: 22 mai 2024.
BRASIL. Resolução CONAMA n° 499, de 06 de outubro de 2020. Dispõe sobre o licenciamento da atividade de coprocessamento de resíduos em fornos rotativos de produção de clínquer. Disponível em: https://conama.mma.gov.br/?option=com_sisconama&task=arquivo.download&id=798. Acesso em: 24 ago 2024.
CLAYTON, T.; R. B. PEARCE, R. B. Rapid chemical analysis of the <2 μm clay fraction using an SEM/EDS technique. Clay Minerals, v. 42, n. 4, 549-562, 2007. https://doi.org/10.1180/claymin.2007.042.4.11. Disponível em: https://is.gd/VkBHdb. Acesso em: 22 mai. 2024
COLETTI, C.; MARITAN L.; CULTRONE, G.; MAZZOLI, C. Use of industrial ceramic sludge in brick production: Effect on aesthetic quality and physical properties. Construction and Building Materials. v. 124, 219-227, 2016. https://doi.org/10.1016/j.conbuildmat.2016.07.096. Disponível em: Disponível em: https://is.gd/O0HlmP. Acesso em: 22 mai. 2024.
COSTA, A. N. C.; SANTOS, J. C.; CARVALHO, J. E. R.; OLIVEIRA, N. S.; MARTINS, T. S. Utilização de pré-tratamento a base de nanotecnologia em linha de pintura da indústria automobilística. Tecnologia em Metalurgia, Materiais e Mineração, v. 9, n. 3, 204-211, 2012. http://dx.doi.org/10.4322/tmm.2012.029. Disponível em: https://is.gd/6QmHwG. Acesso em: 22 mai. 2024.
COSTA, J. S.; AGNOLI, R. D.; FERREIRA, J. Z. Corrosion behavior of a conversion coating based on zirconium and colorants on galvanized steel by electrodeposition. Tecnologia em Metalurgia, Materiais e Mineração, v. 12, n. 2, 167-175, 2015. http://dx.doi.org/10.4322/2176-1523.0852. Disponível em: https://is.gd/IgvyX7. Acesso em: 22 mai. 2024.
DONDI, M.; RAIMONDO, M.; ZANELLI, C. Clays and bodies for ceramic tiles: Reappraisal and technological classification. Applied Clay Science, v. 96, 91–109, 2014. http://dx.doi.org/10.1016/j.clay.2014.01.013. Disponível em: https://is.gd/wBummo. Acesso em: 22 mai. 2024
FINDIK, N. O.; AKYOL, A. A.; SARI, N. Archaeometric analyses of Hasankeyf unglazed ceramics. Mediterranean Archaeology and Archaeometry, v. 14, n. 1, p. 261-271, 2014. Disponível em: https://is.gd/VKUDx9. Acesso em: 22 mai. 2024.
GAZULLA BARREDA, M. F.; EDO, R. F.; CORDERO, M. O.; VAQUER, M. J. V. Determination of minor and trace elements in geological materials used as raw ceramic materials. Boletín de la Sociedad Española de Cerámica y Vidrio, v. 55, 185–196, 2016. http://dx.doi.org/10.1016/j.bsecv.2016.06.003. Disponível em: https://is.gd/Yjvzui. Acesso em: 22 mai. 2024.
HASAN, Md. A.; HASHEM, Md. A.; PAYEL, S. Stabilization of liming sludge in brick production: A way to reduce pollution in tannery. Construction and Building Materials. v. 314, part A, 125702, 2022. https://doi.org/10.1016/j.conbuildmat.2021.125702. Disponível em: https://is.gd/UtjQCa. Acesso em: 22 mai. 2024.
JUEL Md. A. I.; MIZAN A.; AHMED T. Sustainable use of tannery sludge in brick manufacturing in Bangladesh. Waste Management, v. 60, 259-269, 2017. https://doi.org/10.1016/j.wasman.2016.12.041. Disponível em: https://is.gd/MgitIz. Acesso em: 22 mai. 2024.
LIMA, L. K. S.; SILVA, K. R.; MENEZES, R. R.; SANTANA, L. N. L.; LIRA, H. L. Microstructural characteristics, properties, synthesis and applications of mullite: a review. Cerâmica, v. 68, 126-142, 2022. http://dx.doi.org/10.1590/0366-69132022683853184. Disponível em: https://is.gd/1qWql0. Acesso em: 22 mai. 2024
LÓPEZ-JUVINAO, D. D.; TORRES-USTATE, L. M.; MOYA-CAMACHO F. O. Tecnologías, procesos y problemática ambiental en la Minería de arcilla. Investigación e Innovación en Ingenierías, v. 8, n. 2, 20-43, 2020. https://doi.org/10.17081/invinno.8.2.3857. Disponível em: https://is.gd/obHj3b. Acesso em: 22 mai. 2024.
MACEDO, R. S. MENEZES, R. R.; NEVES, G. A.; FERREIRA, H. C. Estudo de argilas usadas em cerâmica vermelha. Cerâmica, v. 54, 411-417, 2008. https://doi.org/10.1590/S0366-69132008000400005. Disponível em: https://is.gd/QMUQdE. Acesso em: 22 mai. 2024.
MARTÍNEZ-GARCÍA, C.; ELICHE-QUESADA, D.; PÉREZ-VILLAREJO, L.; IGLESIAS-GODINO, F. J.; CORPAS-IGLESIAS, F. A. Sludge valorization from wastewater treatment plant to its application on the ceramic industry. Journal of Environmental Management, v. 95, supplement, S343-S348, 2012. https://doi.org/10.1016/j.jenvman.2011.06.016. Disponível em: https://is.gd/WW40Bp. Acesso em: 22 mai. 2024.
METCALF, L.; EDDY. H. P. Tratamento de Efluentes e Recuperação de Recursos, 5. ed. São Paulo: McGraw Hill Brasil, 2015.
MUNIR, M. J; KAZMI, S. M. S.; WU, Y.; HANIF, A.; KHAN, M. U. A. Thermally efficient fired clay bricks incorporating waste marble sludge: An industrial-scale study. Journal of Cleaner Production, v. 174, 1122-1135, 2018. https://doi.org/10.1016/j.jclepro.2017.11.060. Disponível em: https://is.gd/ftttX0. Acesso em: 22 mai. 2024.
MUÑOZ VELASCO, P.; MORALEZ ORTÍZ, M. P.; MENDÍVIL GIRÓ, M. A.; MUÑOZ VELASCO, L. Fired clay bricks manufactured by adding wastes as sustainable construction material – A review. Construction and Building Materials, v. 63, 97-107, 2014. https://doi.org/10.1016/j.conbuildmat.2014.03.045. Disponível em: https://is.gd/MxvKBv. Acesso em: 22 mai. 2024
NUNES C. S.; CASTILLO, A. S.; RAMOS, J. P.; GOMES, L. G.; CARDOSO, F. J. B.; VILHENA, K. S. S. Potential of unprocessed residue from obtaining silicon metal as an adsorbent of methyl red dye. Matéria, v.28, n.1, SP, 2023. https://doi.org/10.1590/1517-7076-RMAT-2023-0018. Disponível em: https://is.gd/dqrHfr. Acesso em: 22 mai. 2024
OLIVEIRA, E. M. S.; HOLANDA, J. N. F. Influência da adição de resíduo (lodo) de estação de tratamento de águas nas propriedades e microestrutura de cerâmica vermelha. Cerâmica, v. 54, 167-173, 2008. https://doi.org/10.1590/S0366-69132008000200006. Disponível em: https://is.gd/kvdc1W. Acesso em: 22 mai. 2024.
ONOFRE, S. B; ABATTI, D.; TESSARO, A. A. Propriedades físico-químicas e microbiológicas do lodo de esgoto produzido pela estação de tratamento de esgoto (ETE) de Toledo – Paraná – Brasil. In: Congresso Brasileiro de Gestão Ambiental, 4., 2015, Porto Alegre Anais... Porto Alegre: IBEAS, 2015. p. 1-6. Disponível em: https://is.gd/1s3y95. Acesso em: 22 mai 2024. – Instituto Brasileiro de Estudos Ambientais
PARENTE, I. M. S.; LYRA, G. P.; BUENO, C.; TONIN, F. G.; ROSSIGNOLO, J. A. Holistic evaluation of ceramic clay properties with Sargassum spp. ash replacement. Construction and Building Materials, v. 435, 136680, 2024. https://doi.org/10.1016/j.conbuildmat.2024.136680. Disponível em: https://is.gd/J98JTP. Acesso em: 22 mai. 2024
PTÁČEK, P.; FRAJKOROVÁ, F.; ŠOUKAL, F.; OPRAVIL, T. Kinetics and mechanism of three stages of thermal transformation of kaolinite to metakaolinite. Powder Technology, v. 264, p. 439–445, 2014. http://dx.doi.org/10.1016/j.powtec.2014.05.047. Disponível em: https://is.gd/y0y5kf. Acesso em: 22 mai. 2024
QUANTUM ANALYTICS. XRD or XRF – Which technique is best for material characterization? Disponível em: https://is.gd/KX6UnG. Acesso em: 22 mai. 2024.
RICHTER, C. A. Tratamento de lodos de estações de tratamento de água. São Paulo: Edgar Blucher, 2001.
RODRIGUES, L. R.; DELLA SAGRILLO, V. P.; REIS, A. S. Efeito das condições de queima na estabilização de um resíduo rico em CaCO3 em massa argilosa. Cerâmica, v. 66, 314-320, 2020. https://doi.org/10.1590/0366-69132020663792906. Disponível em: https://is.gd/TZTGBf. Acesso em: 22 mai. 2024
SAVAZZINI-REIS, A.; DELLA-SAGRILLO, V. P.; VALENZUELA-DIAZ, F. R. Caracterização e propriedades cerâmicas de argilas usadas em cerâmica vermelha no estado do Espiríto Santo. In: CBECiMat – Congresso Brasileiro de Engenharia e Ciência dos Materiais, 22., 2016, Natal. Anais... Fortaleza: UFC/IPEN, 2016. p. 29-39. Disponível em: https://is.gd/Dyy6Oe. Acesso em: 22 mai. 2024.
SIMÓN, D.; BATTISTESSA, C. P.; ARDUZZO, D. C.; GASS, S.; CRISTÓBAL, A. Valorization of sludge from the effluent treatment of the dairy industry as clay substitutes in building bricks. Construction and Building Materials, v. 307, 124955, 2021. https://doi.org/10.1016/j.conbuildmat.2021.124955. Disponível em: https://is.gd/Nra91Z. Acesso em: 22 mai. 2024.
SISINNO, C. L. S. Disposição em aterros controlados de resíduos sólidos industriais não-inertes: avaliação dos componentes tóxicos e implicações para o ambiente e para a saúde humana. Cadernos de Saúde Pública, v. 19, n. 2, 369-374, 2003. https://doi.org/10.1590/S0102-311X2003000200003. Disponível em: https://is.gd/AOtUkb. Acesso em: 22 mai. 2024.
SUTCU, M.; GENCEL, O.; ERDOGMUS, E.; KIZINIEVIC, O.; KIZINIEVIC, V.; KARIMIPOUR, A.; VELASCO. P. M. Low cost and eco-friendly building materials derived from wastes: Combined effects of bottom ash and water treatment sludge. Construction and Building Materials, v. 324, 126669, 2022. https://dx.doi.org/10.1016/j.conbuildmat.2022.126669. Disponível em: https://is.gd/n3e2tS. Acesso em: 22 mai. 2024.
TARTARI, R.; DÍAS-MORA, N.; MÓDENES, A. N.; PIANARO, S. A. Lodo gerado na estação de tratamento de água Tamanduá, Foz do Iguaçu, PR, como aditivo em argilas para cerâmica vermelha. Parte I: Caracterização do lodo e de argilas do terceiro planalto paranaense. Cerâmica, v. 57, 288-293, 2011. https://doi.org/10.1590/S0366-69132011000300006. Disponível em: https://is.gd/xDPaxI. Acesso em: 22 mai. 2024.
TEIXEIRA, S. R; SANTOS, G. T. A.; SOUZA, A. E.; ALESSIO, P., SOUZA, S. A.; SOUZA, N. R. The effect of incorporation of a Brazilian water treatment plant sludge on the properties of ceramic materials. Applied Clay Science, v. 53, n. 4, 561-565, 2011. https://doi.org/10.1016/j.clay.2011.05.004. Disponível em: https://is.gd/CSXzuU. Acesso em: 22 mai. 2024
TURRIÓN, D. MORCILLO, L.; ALLOZA, J. A.; VILAGROSA, A. Innovative Techniques for Landscape Recovery after Clay Mining under Mediterranean Conditions. Sustainability. v. 13, n. 6, 3439, 2021. https://doi.org/10.3390/su13063439. Disponível em: https://is.gd/iTi43g. Acesso em: 22 mai. 2024.
UE. Directiva 2008/98/CE do Parlamento Europeu e do Conselho, de 19 de novembro de 2008, relativa aos resíduos e que revoga certas directivas. L 312/3. Disponível em: https://is.gd/8gMesx. Acesso em: 22 mai. 2024.
UKWATTA, A.; MOHAJERANI, A.; SETUNGE, S.; ESHTIAGHI, N. Possible use of biossolids in fired-clay bricks. Construction and Building Materials, v. 91, 86-93, 2015. https://doi.org/10.1016/j.conbuildmat.2015.05.033. Disponível em: https://is.gd/K2P2dH. Acesso em: 22 mai. 2024.
UKWATTA, A.; MOHAJERANI, A.; ESHTIAGHI, N.; SETUNGE, S. Variation in physical and mechanical properties of fired-clay bricks incorporating ETP biosolids. Journal of Cleaner Production, v. 119, 76-85, 2016. https://doi.org/10.1016/j.jclepro.2016.01.094. Disponível em: https://is.gd/qmSkjj. Acesso em: 22 mai. 2024.
UKWATTA, A.; MOHAJERANI, A. Characterisation of fired-clay bricks incorporating biosolids and the effect of heating rate on properties of bricks. Construction and Building Materials, v. 142, 11-22, 2017. https://doi.org/10.1016/j.conbuildmat.2017.03.047. Disponível em: https://is.gd/CW16og. Acesso em: 22 mai. 2024.
USEPA. U.S. ENVIRONMENTAL PROTECTION AGENCY. Test Method for Evaluating Solid Waste: Physical/Chemical Methods Compendium SW-846: The SW- 846 Compendium. Washington, DC (1986). Disponível em: https://www.epa.gov/hw-sw846/sw-846-compendium. Acesso em: 22 mai. 2024
USEPA. Method 245.7 - Mercury in water by cold vapor atomic fluorescence spectrometry. Revision 2.0 Washington, DC. 1986.
USEPA. Method 300.0 - Determination of Inorganic Anions in Drinking Water by Ion Chromatography. Revision 2.1. Cincinnati, Ohio. 1993.
USEPA. Method 3510C - Separatory funnel liquid-liquid extraction. Revision 3.0. Washington, DC. 1996.
USEPA. Method 3531 (SW 846) - Ultrasonic extraction. Revision 3.0. Washington, DC. 2007.
USEPA. Method 5021 (SW 846) - Volatile organic compounds in various sample matrices using equilibrium headspace analysis. Revision 2.0. Washington, DC.
2014.
USEPA. Method 6010D (SW-846) - Inductively Coupled Plasma-Atomic Emission Spectrometry. Revision 4. Washington, DC. 2014.
USEPA. Method 8081B (SW 846) - organochlorine pesticides by gas chromatography. Revision 2.0. Washington, DC. 2007.
USEPA. Method 8260C (SW 846) - Volatile organic compounds by gas chromatography/ mass spectrometry. Revision 3.0. Washington, DC. 2006.
USEPA. Method 8270D (SW 846) - Semivolatile organic compounds by gás chromatography/mass spectrometry (GC/MS). Revision 4.0. Washington, DC.1998
USEPA. Method 9010C - Total and amenable cyanide: distillation. Revision 3.0. Washington, DC. 2004
USEPA. Method 9034 (SW 846) - Titrimetric procedure for acid-soluble and acid insoluble sulfides. Washington, DC.1996.
USEPA. Method 9045D (SW 846) - Soil and waste pH test. Revision 4.0. Washington, DC. 2004
USEPA. Method 9213 (SW-846): Potentiometric determination of cyanide in aqueous samples and distillates with ion-selective electrode. Washington, DC. 1996.
VIEIRA, G.; SILVA, D. F. Estudo da adição de lodo gerado pela indústria metal mecânica na fabricação de tijolos de cerâmica vermelha. E-tech: Tecnologias para competitividade Industrial, Florianópolis, n. esp. Metalmecânica . v. sv, 69-91, 2012. https://doi.org/10.18624/e-tech.v0i0.220. Disponível em: https://is.gd/oanlEV. Acesso em: 22 mai. 2024.
YARAS, A. Combined effects of paper mill sludge and carbonation sludge on characteristics of fired clay bricks. Construction and Building Materials v. 249, 118722, 2020. https://doi.org/10.1016/j.conbuildmat.2020.118722. Disponível em: https://is.gd/WdWDnv. Acesso em: 22 mai. 2024.
ZHANG, Y. M.; JIA, L. T., MEI, H.; CUI, Q.; ZHANG, P. G.; SUN, Z. M. Fabrication, microstructure and properties of bricks fired from lake sediment, cinder and sewage sludge. Construction and Building Materials, v. 121, 154-160, 2016. https://doi.org/10.1016/j.conbuildmat.2016.05.155. Disponível em: https://is.gd/mEE5Pj. Acesso em: 22 mai. 2024.


